COVID-19 | Influenza Respiratory Illnesses

Respiratory Test Kit ID NOW™ Strep A 2.0 24 Tests CLIA Waived
Positive results detected in as few as 2 minutes, with 99% detected within 3 minutes Enhanced molecular sensitivity eliminates the need for culture confirmation of negative results Simple operation via visual touchscreen Connected results direct to your network More Information ID NOW Strep A 2.0 Assay Package Insert
$1,500.00

Respiratory Test Kit ID NOW™ Influenza A + B 2.0 Influenza A + B 24 Tests per Kit CLIA Waived
Features ID NOW™ Influenza A & B 2, Buy 4, Get 6 Promo Pack (i.e. 6 kits total) Alere I is now ID Now™ Small footprint enables ease of use at point of care Simple operation via visual touchscreen Highly accurate results in 13 minutes Integrated pipette eliminates manual pipetting for direct samples Standardized respiratory trio (Influenza A & B, RSV, Strep) across all sites of care Bi-directional connectivity to industry leading RALS® platform facilitates satellite testing and remote management CLIA COMPLEXITY: WAIVED - For Direct Nasal Swabs (Not Eluted in Viral Transport Media) Only
$1,500.00

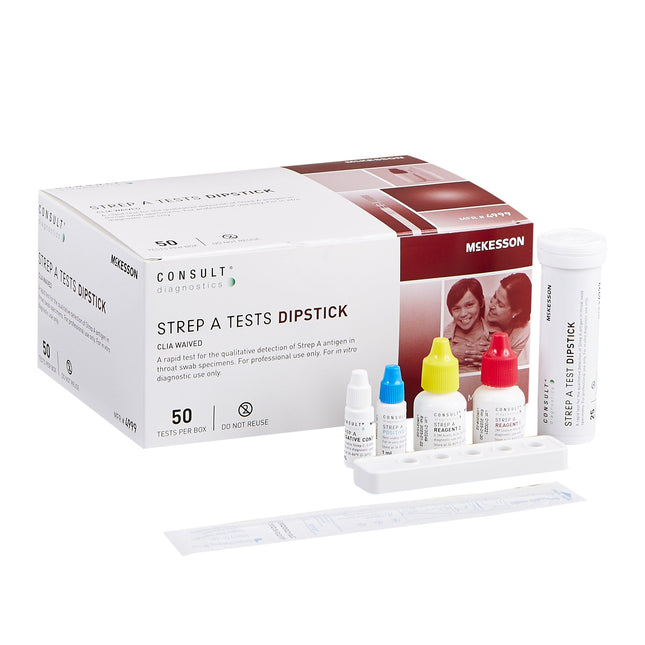
MCI Supplies- Medical Supply Company Respiratory Test Kit McKesson Consult™ Strep A Test 50 Tests CLIA Waived
McKesson Consult™ Strep A Dipstick Test A rapid test for the qualitative detection of Strep A antigen in throat swab specimens CLIA waived and easy to use Contents: 50 Dipsticks (2 vials of 25), Disposable extraction test tubes, Sterile swabs, Reagent 1 (10 mL; 2M Sodium Nitrite), Reagent 2 (10 mL; 0.2M Acetic Acid), Positive control (1 mL; Non-viable Strep A; 0.09% NaN3), Negative control (1 mL; Non-viable Strep C; 0.09% NaN3), Instructional insert, Workstations. Results in five minutes. Accurate – 97% sensitivity and 95% specificity External positive and negative quality controls provided For in vitro diagnostic use only 24-month shelf life Not Made with Natural Rubber Latex Packaged: 50 Tests Per Box
$84.18

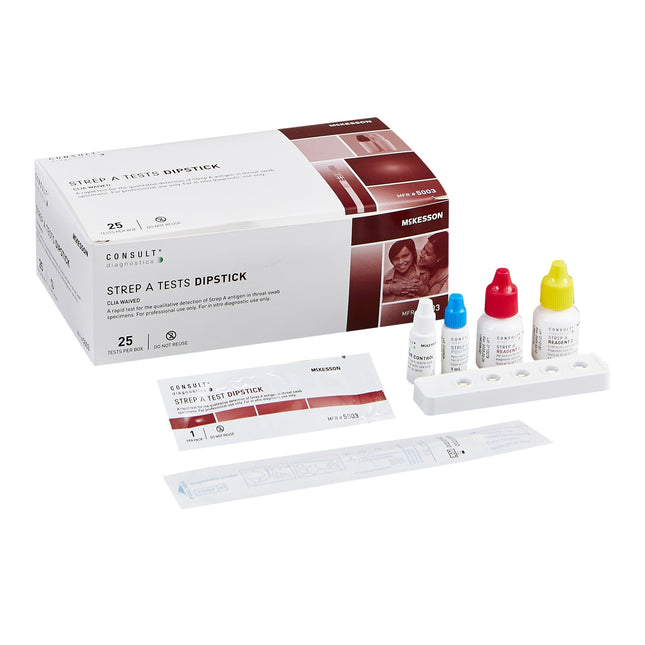
MCI Supplies- Medical Supply Company Respiratory Test Kit McKesson Consult™ Strep A Test 25 Tests CLIA Waived
McKesson Consult™ Strep A Dipstick Test A rapid test for the qualitative detection of Strep A antigen in throat swab specimens CLIA waived and easy to use Contents: 25 Dipsticks (in sealed pouches), Disposable extraction test tubes, Sterile swabs, Reagent 1 (10 mL; 2M Sodium Nitrite), Reagent 2 (10 mL; 0.2M Acetic Acid), Positive control (1 mL; Non-viable Strep A; 0.09% NaN3), Negative control (1 mL; Non-viable Strep C; 0.09% NaN3), Instructional insert, Workstations Results in five minutes Accurate – 97% sensitivity and 95% specificity External positive and negative quality controls provided For in vitro diagnostic use only 24-month shelf life Not Made with Natural Rubber Latex Packaged: 25 Tests Per Box
$50.00

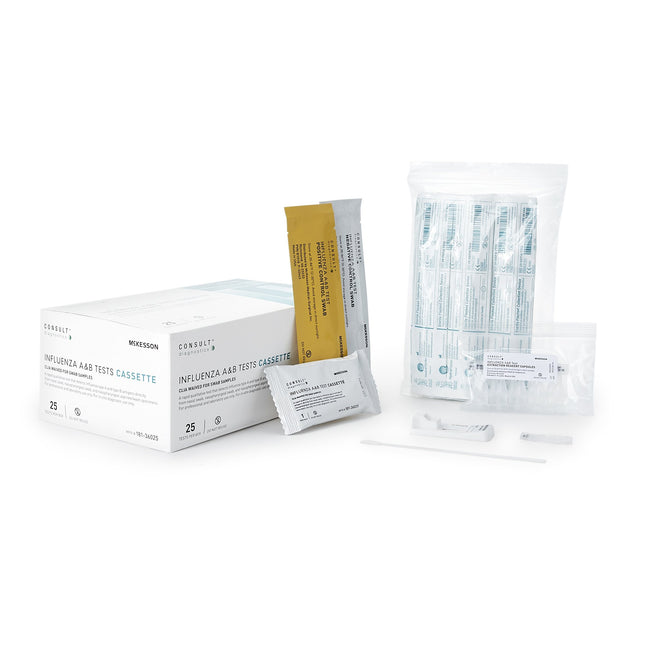
MCI Supplies- Medical Supply Company Respiratory Test Kit McKesson Consult™ Influenza A + B 25 Tests CLIA Waived
McKesson Consult™ Influenza A & B Tests Rapid qualitative test that detects Influenza type A and type B antigens directly from nasal swab, nasopharyngeal swab, and Nasopharyngeal aspirate / wash specimens Color-coded control swab packaging for easy positive/negative identification Results in under 15 minutes CONTENTS: 25 Test Cassettes, 25 Sterile Swabs, 25 Extraction Reagent Capsules, 1 Positive Control Swab, 1 Negative Control Swab, 1 Procedure Card, 1 Instructions for Use CLIA Complexity: Moderate Complexity when used with Nasopharyngeal Wash/Aspirate Samples- CLIA Waived when used with Nasal and Nasopharyngeal Swabs For aspirate samples, kit sold separately (MFR#: 181-36026) For professional and laboratory use only For in vitro diagnostic use only Not Made with Natural Rubber Latex Packaged: 25 Tests Per Box
$310.58

Respiratory Test Kit ID NOW™ COVID-19 2.0 24 Tests CLIA Waived
FDA-cleared ID NOW™ COVID-19 2.0 detects SARS-CoV-2 in 6–12 minutes with the option to add on the ID NOW™ Influenza A & B 2 test without collecting another sample Product ships with minimum 30 days dating Highly accurate, rapid molecular COVID-19 test The ID NOW platform uses isothermal technology to provide molecular results faster than PCR for more timely and informed clincial decisions CLIA-waived; requires minimum training with easy on-screen video-guided operation and no manual pipetting required ID NOW™ Platform gives you the power to decide which tests to run based on patient presentation, circulating prevalence and seasonality of infectious diseases Easily add on Flu A & B testing based on clinical necessity, supporting diagnostic stewardship Diagnostic Stewardship: Right tests at the right time for optimal care and fewer unnecessary tests Room Temperature Storage For use with the ID NOW Instrument
$1,180.80

MCI Supplies- Medical Supply Company Respiratory Test Kit BinaxNOW™ Professional Use COVID-19 Ag 40 Tests
BinaxNOW COVID-19 Ag Card is only for use under the Food and Drug Administration’s Emergency Use Authorization: https://www.fda.gov/medical-devices/emergency-situations-medical-devices/emergency-use-authorizations Testing is limited to laboratories certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA), 42 U.S.C. §263a, that meet the requirements to perform moderate, high or waived complexity tests; this test is authorized for use at the Point of Care (POC), i.e., in patient care settings operating under a CLIA Certificate of Waiver, Certificate of Compliance, or Certificate of Accreditation Product ships with minimum 60 days dating This test is authorized for use with direct anterior nasal (nares) swab samples from individuals with symptoms of COVID-19 within the first seven days of symptom onset when tested at least twice over three days with at least 48 hours between tests; this test is also authorized for use with direct anterior nasal (nares) swab samples from individuals with or without symptoms or other epidemiological reasons to suspect COVID-19 when tested at least three times over five days with at least 48 hours between tests Laboratories within the United States and its territories are required to report all results to the appropriate public health authorities Positive results indicate the presence of viral antigens, but clinical correlation with patient history and other diagnostic information is necessary to determine infection status Positive results do not rule out bacterial infection or co-infection with other viruses Negative results should be treated as presumptive and confirmation with a molecular assay, if necessary, for patient management, may be performed Negative results do not rule out SARS-CoV-2 infection and should not be used as the sole basis for treatment or patient management decisions, including infection control decisions The BinaxNOW COVID-19 Ag Card is intended for use by medical professionals or trained operators who are proficient in performing rapid lateral flow tests Sensitivity (PPA) 84.6% (entire population) Sensitivity (PPA) 95.6% (those with PCR cycle threshold [Ct] < 33) Specificity (NPA) 98.5% Supplemental data indicates that antigen tests have demonstrated positive percent agreement as high as 100% for symptomatic patients (3 serial tests, 2 days after first PCR positive) and as high as 88.9% for asymptomatic patients (3 serial tests, 6 days after first PCR positive) Onboard extraction allows the swab to be directly inserted into the test card Visually read results in 15 minutes - no instrument required The BinaxNOW COVID-19 Ag Card does not differentiate between SARSCoV and SARS-CoV-2NOTE: THIS PRODUCT IS NON-RETURNABLE
$375.00